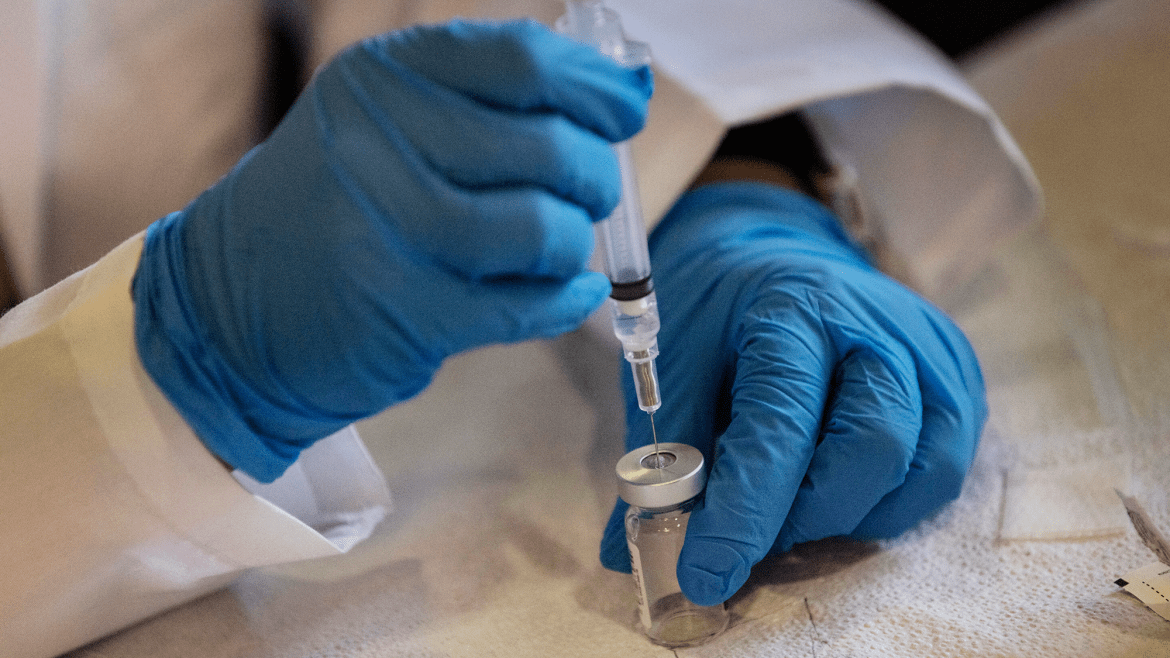
There’s no ambiguity about who Vyleesi is for. The prescription drug, commonly referred to as female Viagra, boosts the libido of women experiencing hypoactive-sexual-desire disorder, a condition in which lack of interest in sex causes distress. When Vyleesi was approved in 2019, an FDA official announced that it was evidence of the agency’s “commitment to protect and advance the health of women.”
But since then, female Viagra seems to have found an unexpected market: men. On the Reddit forum SexOnDrugs—which chronicles, you guessed it, people’s sexual escapades while on various prescription and illicit drugs—guys talk about taking the drug even if they have no apparent issues with sexual drive or performance. “Everything feels richer,” noted one man, who compared taking the drug to adding butter to food. Another claimed that he’d had sex with his wife “about 30 times over 20 hours.” (The drug, however, does frequently make people queasy: “The nausea hit me so hard that sex wasn’t even a thought,” another man reported.)
Vyleesi has never been approved for men. Some clinics advertise that they’ll prescribe the drug to men off-label, but even that is often not necessary for men to get ahold of it. Vyleesi is now readily available without a prescription. Many online retailers sell vials of the drug under the guise that they are for “research use only” and not for human consumption—a disclaimer that technically makes the drugs legal.
Americans are relying on this technicality to get hold of all kinds of drugs—some that, like Vyleesi, are supposed to require a prescription and others that aren’t even approved in the United States at all. Athletes are taking a banned horse-racing drug to speed up their recovery. Beauty influencers are shooting something known as the “Barbie drug” up their nose to get a better tan. My colleague Sarah Zhang recently described the process of buying retatrutide, an unapproved obesity drug, as “just like ordering socks.”
[Read: I bought ‘GLP-3’]
Not all experimentation requires scouring the gray market. Online pharmacies staffed by licensed doctors now regularly prescribe “personalized” medications that include an FDA-approved medication paired with additional experimental additives. One company, BlueChew, sells an erectile-dysfunction pill that contains the active ingredients in Viagra and Cialis, in addition to other chemicals that the company claims improve sex. These products are made by compounding pharmacies, which are not regulated by the FDA for safety, quality, or efficacy.
Taking experimental drugs is nothing new. Sigmund Freud frequently used cocaine to treat his depression; athletes have doped for decades; biohackers have long taken pills in hopes of becoming more efficient at work. What is new, however, is the scale, accessibility, and uptake of faddish pharmaceuticals. Now everyone is a biohacker—or at least anyone can be.
Few experimental drugs have become as popular as peptides—a class of compounds that mimic existing hormones in the body. Peptides have developed a cult following among fitness enthusiasts, looks-maxxers, and Silicon Valley types, who believe that the drugs can boost muscle gain, increase focus, and lead to better skin. On a recent episode of his podcast, Joe Rogan urged Ben Affleck and Matt Damon to use the peptides BPC-157 and TB-500—commonly known as the “Wolverine stack”—to speed up recovery should they ever get injured.
The P in GLP-1 stands for peptide. The success of Ozempic and other FDA-approved drugs for weight loss and diabetes “opened a lot of eyes,” Dave Asprey, a longevity influencer who credits himself with starting the biohacking movement, told me. “People started asking what else is out there.” And there is, it turns out, a ton out there. One online vendor of unapproved peptides that are not intended for human consumption carries dozens of drugs on its website.
GLP-1s are also a big part of the reason Americans are now experimenting with unregulated drugs made by compounding pharmacies. Legally, these businesses are supposed to make custom versions of drugs for people who can’t take an FDA-approved drug, or when an FDA-approved drug is unavailable because of a shortage. Roughly a decade ago, entrepreneurs realized that they could use compounding pharmacies to make nominally customized versions of FDA-approved medicines and sell them to the masses via the internet. Hims pioneered the strategy, selling hair spray loaded with finasteride, an FDA-approved hair-loss drug previously available only in pill form. Now telehealth companies offer compounded drugs for sexual health, hair growth, skin care, and especially weight loss. Sales of these obesity-drug dupes have become an enormous business; both Hims and Ro ran Super Bowl ads touting their offerings.
[Read: GLP-1 envy was just the beginning]
Some of this might seem like a positive development for American medicine. In the cases of weight loss and erectile dysfunction, more patients are getting the care they want or need. I personally experimented with compounded GLP-1s because of the high cost of the real thing. But again, these drugs are not made with any government oversight. Consider Musely, a company that sells skin cream for dark spots. The cream contains three times the amount of the active ingredient hydroquinone, which is FDA-approved, that you’d get in the traditional version of the drug—despite the fact that regulators have warned that hydroquinone may cause skin discoloration. (Musely did not respond to a request for comment.)
Experimental drugs that lack any FDA-approved ingredients can be even more questionable. Although anecdotal evidence abounds about the benefits of the “Wolverine stack” and certain other peptides, most haven’t been researched enough to prove they actually work. “We don’t even know what’s in these bottles,” Christopher Robertson, a Boston University law professor who studies the FDA, told me.
The modern FDA exists to ensure that pharmaceuticals are safe. But the agency has failed to stop the boom of unregulated, potentially dangerous drugs. It has gone after some companies for selling research chemicals after determining that the drugs were, in fact, being sold for human consumption, but the agency is playing a game of whack-a-mole. Dozens of websites selling BPC-157 can be found via a quick Google search. Shutting down compounding pharmacies operating on the gray market would likely be even more difficult, and prompt protracted court battles.
It’s easy to take the FDA for granted. In the 1960s, thalidomide, a drug marketed for morning sickness, left children around the world with irreversible birth defects; the United States avoided such a fate thanks to the FDA’s oversight. Due to the thalidomide scare, America began to require drugs to be proved not only safe but also effective.
Robert F. Kennedy Jr., who oversees the FDA and other major health agencies as the secretary of Health and Human Services, knows this history. During his confirmation hearing, Kennedy name-checked Frances Oldham Kelsey, the FDA official who refused to approve thalidomide in the U.S., as an example of how scientists must follow the data and question science. Yet Kennedy seems poised to make it even easier for Americans to get certain unregulated drugs. On Rogan’s podcast late last month, Kennedy teased that he will soon act to make roughly a dozen peptides “more accessible,” in line with his penchant for medical freedom. He also revealed that he is a “big fan” of certain peptides and has used them himself to help with injuries. (An HHS spokesperson did not respond to a request for comment.)
Just as Americans’ appetite for experimental drugs reaches new heights, the FDA’s capacities to ensure the safety of America’s medicines seem to be diminishing.