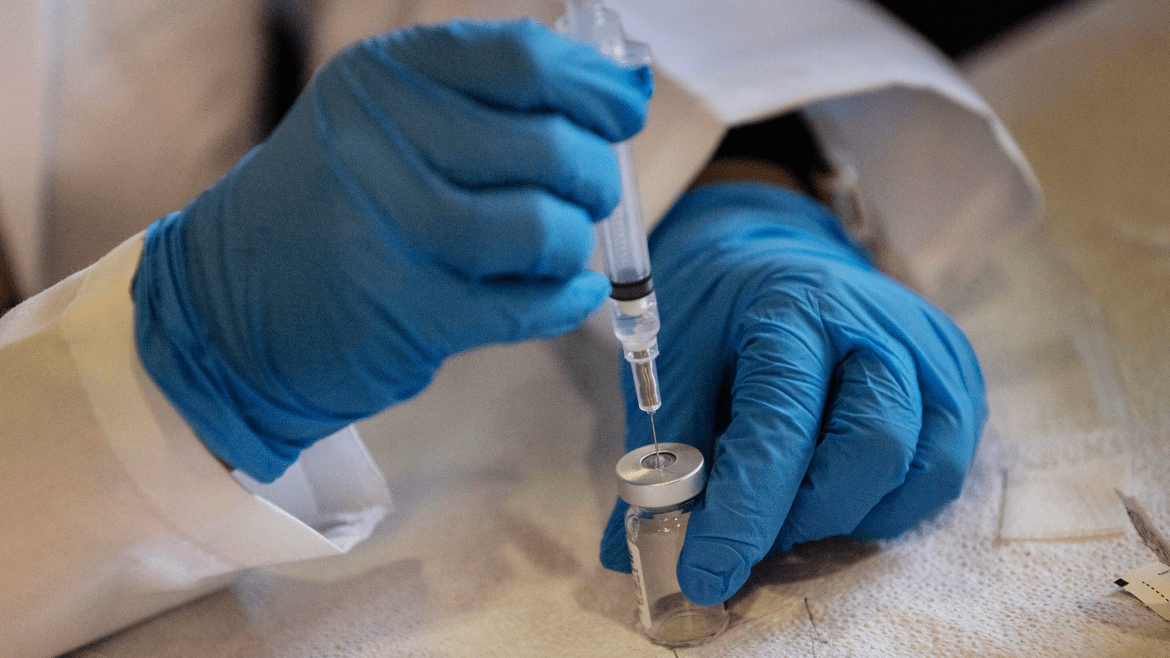
On Monday, a federal judge issued a preliminary ruling with a harsh reprimand for the Trump administration: You’ve done this vaccine stuff all wrong.
The Trump administration likely broke the law, the judge’s 45-page decision argued, when it dismissed and abruptly reconstituted the CDC’s expert vaccine-advisory panel last June, stacking the committee with members who have aggressively questioned the safety of vaccines. Top health officials also probably acted illegally, the ruling said, when they made sweeping alterations to the nation’s childhood-immunization schedule in January, without the input of their own, remade panel.
If the decision becomes final, it stands to all but wipe away a year’s worth of vaccine-policy change at the CDC. The judge’s ruling calls for staying “all votes taken” by the panel since Health and Human Services Secretary Robert F. Kennedy Jr. remade it—essentially resetting the United States’ vaccine-policy clock to early 2025. But the ruling remains preliminary, and the Trump administration has already hinted at its intent to appeal. Andrew Nixon, the deputy assistant secretary for media relations at HHS, did not respond to a request for comment, but he told reporters this week that “HHS looks forward to this judge’s decision being overturned just like his other attempts to keep the Trump administration from governing.”
Since the Trump administration took office, states and professional medical societies have broken with the CDC, vaccine recommendations have splintered, and trust in federal health agencies, especially in their vaccine advice, has plunged. This latest ruling shifts the nature of the United States’ vaccine chaos, but the turmoil is far from done: Lawyers and judges could be arguing for months over who has the authority to set U.S. vaccine policy.
As things stand, the CDC’s national immunization schedule—and the primary committee that shapes it—is in a kind of purgatory. The current roster of the panel, the Advisory Committee on Immunization Practices (ACIP), should never have come to exist at all, the judge wrote, because it violated the Administrative Procedure Act: When Kennedy fired all 17 of the panel’s former members last spring and replaced them with a more vaccine-skeptical group, he eschewed the “rigorous screening that had been the hallmark of ACIP member selection for decades.” Kennedy’s ACIP, the ruling says, may also fail to fulfill a requirement of the Federal Advisory Committee Act, which calls for the membership of such panels to have “balanced” points of view. The ruling temporarily suspended the appointment of 13 of the 15 current ACIP members, most of whom lack “meaningful experience in vaccines,” the judge wrote. (The other two members were appointed in late February, after the plaintiffs filed their motion, and so were excluded from the decision.) As a result, the panel has been forced to postpone a meeting originally scheduled for this week.
All of that effectively leaves the U.S. without the advisory group that has most influentially shaped American vaccine policy for the past 60 years. “There is no functioning ACIP as of now,” Richard Hughes IV, a lawyer for the plaintiffs, told me. The nation has never been so suddenly stripped of its vaccine advisers, with no backup plan. Experts told me they’re unsure who, if anyone, will advise the CDC on its recommendations for vaccines in the coming months, when the agency might need to weigh in on brand-new immunizations against diseases that have so far lacked them, as well as as this fall’s slate of vaccines against COVID and flu, which are typically reformulated annually to keep up with viral evolution.
In theory, Kennedy or the CDC director—a position temporarily held by Jay Bhattacharya, who also leads the National Institutes of Health—could simply make a unilateral decision about future vaccine recommendations. But the judge also faulted Jim O’Neill, the CDC’s previous acting director, for doing exactly that in January, when O’Neill signed a memo announcing major changes to the agency’s immunization schedule that downgraded recommendations for several vaccines at once, without seeking ACIP’s advice. “The CDC cannot simply bypass ACIP in altering the immunization schedules,” the judge wrote. And because part of the judge’s ruling criticized the hurried way in which the current ACIP was assembled, Kennedy might have a difficult time summoning a fresh panel to replace most of his picks on short notice.
In the lawsuit that led to this week’s ruling, the plaintiffs—which include major medical groups, such as the American Academy of Pediatrics and the Infectious Diseases Society of America—challenged three of ACIP’s votes over the past year in particular. Last summer, on the advice of an anti-vaccine activist, the panel voted to stop recommending the use of flu vaccines containing thimerosal, a preservative with a proven safety record. In September, it opted to downgrade what was once a universal recommendation for COVID vaccines to guidance that Americans first consult a health-care provider. Then, in December, it voted to rescind a long-standing recommendation that all newborns receive a hepatitis-B vaccine, a change that health experts fear could cause cases of severe liver complications to skyrocket.
But if the ruling actually stays “all votes” taken by Kennedy’s ACIP, it could also undo moves the panel has made that are very much in line with what other versions of ACIP have or would have done, Dorit Reiss, a vaccine-law expert at UC Law San Francisco, told me. For example, the committee recommended the 2025–26 flu vaccines for all Americans six months and older, as it has done in years prior. It also recommended a new monoclonal antibody that can protect babies against RSV, the leading cause of infant hospitalization in the U.S., and qualified the shot for the Vaccines for Children program, which helps immunizations reach underinsured families. If those decisions are canceled, coverage for families that have relied on those shots could be compromised.
For now, the decision may accomplish more symbolically than it does practically. It holds that evidence and proper procedure are key to formulating vaccine policy in the United States. But the actions of both the Trump administration and the judge suggest that the government is still conflicted over just how crucial ACIP is. Kennedy and his allies have taken great care to fill the panel with people whose views align with theirs, a tacit endorsement of ACIP’s importance; at the same time, they have made some of their largest modifications to the national immunization schedule without the committee’s input. The judge’s ruling, too, emphasized the influence of ACIP, noting that the committee is the only body that can determine which immunizations qualify under Vaccines for Children—even as it has frozen ACIP’s ability to act for the foreseeable future.
The ruling does not erase, or even much lessen, the deep sense of unease over vaccine decision making in this country that has resulted from the past year of haphazard changes; if anything, a prolonged legal battle could further diminish public trust, Grace Lee, a former chair of the advisory committee and a pediatrician at Stanford, told me. Kennedy, O’Neill, and other administration officials have repeatedly cited a goal of restoring public trust when modifying the nation’s vaccine recommendations. But one recent poll found that trust in the CDC had dropped since the start of the second Trump administration, and that fewer than half of respondents now trust the agency at least “a fair amount” to provide reliable information on vaccines. Several professional medical societies, including the American Academy of Pediatrics, have broken away from CDC vaccine advice; “no serious health-care provider looks to ACIP anymore for advice,” Noel Brewer, a vaccine-behavior expert at the University of North Carolina’s Gillings School of Global Public Health whom Kennedy dismissed from ACIP last year, told me. Dozens of states have declared that they’re no longer following the CDC on vaccines, either. Americans, in effect, have been left with all too many options for whom to listen to about vaccines.
Even if court proceedings eventually revert the country’s immunization schedule to a version of what it was about a year ago—and even if the administration reverts ACIP to its typical process of rigorously vetting members—the U.S. will still be far from its previous vaccine status quo. Vaccination depends not just on the governing bodies that issue recommendations about shots, but also on Americans’ willingness to heed that advice—a far harder shift to reset.